Air Care / B Pod Series: Intravenous Fluid Resuscitation
/HISTORY OF PRESENT ILLNESS:
The patient is a male in his 70s who presents to the emergency department (ED) with dyspnea and hypoxemia. He has a past medical history of atrial fibrillation on rivaroxaban, diabetes mellitus type 2, and rheumatoid arthritis on prednisone.
Approximately 48 hours prior to this presentation, the patient was admitted to the hospital after being brought in by a concerned family member who found him disoriented at home with a diarrheal illness. On the patient’s initial presentation to the ED, he was found to be febrile and in atrial fibrillation with rapid ventricular rate. While conversant, he was mildly confused with clear lungs and a soft, non-tender abdomen. The patient was resuscitated with intravenous fluids and empiric antibiotics, and was admitted to the medical floor where he remained on a continuous infusion of 0.9% normal saline (NS). He was briefly transferred to the intensive care unit (ICU) after becoming hypotensive, where the fluid rate was doubled and he received additional boluses of NS. Over the next 24 hours, his hemodynamics stabilized and his diarrhea resolved. An infectious work up revealed no bacterial source for his symptoms, antibiotics were discontinued, and he was discharged after a 36-hour hospital stay.
Less than 12 hours following discharge, the patient is brought back to the ED by emergency medical services who found him to be hypoxic to 81% on room air. Upon arrival to the ED, the patient is unable to provide any history due to respiratory distress.
PAST MEDICAL HISTORY:
Atrial fibrillation
PAST SURGICAL HISTORY:
Total Knee Arthroplasty
MEDICATIONS:
Rivaroxaban, Carvedilol, Metformin, Lisinopril, Atorvastatin, Prednisone
ALLERGIES: No known drug allergy
VITALS:
HR 170 bpm, BP 156/99 mmHg, RR 34 breaths/min, SpO2 78% on 10 L/min via nasal cannula
PHYSICAL EXAM:
The patient is a diaphoretic, ill-appearing male who appears his stated age. His neck exam has jugular venous distension to the angle of the mandible with 2+ lower extremity edema in cool lower extremities with 1+ pulses bilaterally. The patient is in moderate respiratory distress, with suprasternal retractions and on auscultation there are diffuse inspiratory crackles and intermittent expiratory wheezes. The patient is tachycardic with an irregularly irregular rhythm without appreciable murmur.
DIAGNOSTIC WORK UP:
WBC – 16, Hgb – 14.3, Hct – 45.1, Platelets – 169
Na – 146, K – 4.4, Cl – 117, CO2 – 10, Anion gap – 19, Glucose – 223, BUN – 17, Cr – 1.2
Protein – 7.2, Albumin – 4.0, Alk phos – 67, ALT – 87, AST – 109, Tbili – <0.2
Troponin – <0.01 Pro-BNP – 2,132
INR – 1.6
VBG – pH 7.13, pCO2 29, pO2 61, HCO3 9, base excess –19
Lactate – 4.8
Urinalysis – SG > 1.030, blood moderate, pH 6.0, protein 30, nitrite negative, leukocyte esterase negative
ECG – atrial fibrillation with rapid ventricular response, rate 151, left axis deviation, T wave inversions in 1 and aVL, minimal ST depression in V4-V6
Imaging:
Chest radiograph – diffuse bilateral air space opacities, more prominent in the right base. Consistent with pulmonary edema, although consolidation cannot be excluded in the right middle and lower lobe.
HOSPITAL COURSE:
The patient was immediately placed on non-invasive positive pressure ventilation with an FiO2 of 100%, given a bolus of furosemide, and placed on a nitroglycerin infusion at an escalating dose. Given the concern for possible sepsis, specifically legionella pneumonia with recent diarrheal illness, broad spectrum antibiotics were given. The patient’s minute ventilation was calculated to be approximately 20 liters per minute. Upon admission to the ICU, his minute ventilation remained unchanged, although his FiO2 was weaned to 50%. Cardiology was consulted for acute decompensated heart failure, and the patient was started on infusions of both amiodarone and furosemide. A transthoracic echocardiogram showed a newly reduced left ventricular ejection fraction (EF) of 15-20% with mild right ventricular dysfunction and no valvular pathology. Over the course of 24 hours, the patient was diuresed four liters and was weaned off positive pressure ventilation to nasal cannula. Intravenous amiodarone and furosemide were transitioned to an oral regimen. Urine legionella assay was negative. Blood cultures returned as no growth after 48 hours. He was discharged and referred for implantable cardioverter-defibrillator placement given his severely reduced EF.
DISCUSSION:
This elderly gentleman presented in respiratory distress with an anion gap metabolic acidosis, non-anion gap metabolic acidosis, and a respiratory acidosis. Although there are several pathophysiologic processes at play, this patient’s clinical course may have been impacted by the fluid therapy he was given during his initial inpatient stay, during which he received approximately seven liters of NS over the first 24 hours of his hospitalization. Throughout his initial admission his bicarbonate decreased from 15 to 12, while his sodium and chloride went from 129 and 99 to 146 and 117, respectively. His large volume, chloride-liberal fluid administration may have played a role in the short span between admissions.
Intravenous fluid (IVF) is one of the most commonly provided treatments in the ED and inpatient settings. Nearly 30 million patients receive IVF annually in the United States alone (1). It is so routine that physicians may forget that the potential benefits of fluid resuscitation can be outweighed based on fluid choice and the volume prescribed. The remainder of this discussion will focus on the options when choosing IVF, including the composition of each fluid and the available data regarding the use of each in particular patient populations.
INTRAVENOUS FLUID OPTIONS
COLLOID V. CRYSTALLOID:
At the most fundamental level, fluids can be divided into crystalloid and colloid agents. Crystalloids are an array of dissolved salts designed to mirror the osmotic composition of plasma. Colloids are comprised of larger molecules, such as albumin or synthetic carbohydrates, aiming to hold volume in the intravascular compartment through the exertion of oncotic force. Both options were designed to achieve the same goal: optimize intravascular volume and restore perfusion in patients suffering from shock. In practice, large scale trials have led many to avoid colloids as some studies have demonstrated evidence of harm with use of colloids, including increased need for renal replacement therapy and even mortality (2,3). Albumin, while safe to use in most critically ill non-trauma patients, has not been found to improve mortality or patient-centered outcomes, such as ventilator or ICU-free days (4,5). In 2013, the Colloids vs. Crystalloids for the Resuscitation of Critically Ill (CRISTAL) trial, a large, multicenter, randomized investigation, found no difference in 28-day mortality between groups of hypovolemic shock patients who received crystalloid versus colloid resuscitation after being admitted to the ICU (6). In the face of large trials showing no benefit to increased harm, and a hefty price tag compared to crystalloid options, colloids are rarely utilized as a primary mode of resuscitation.
CRYSTALLOID V. CRYSTALLOID:
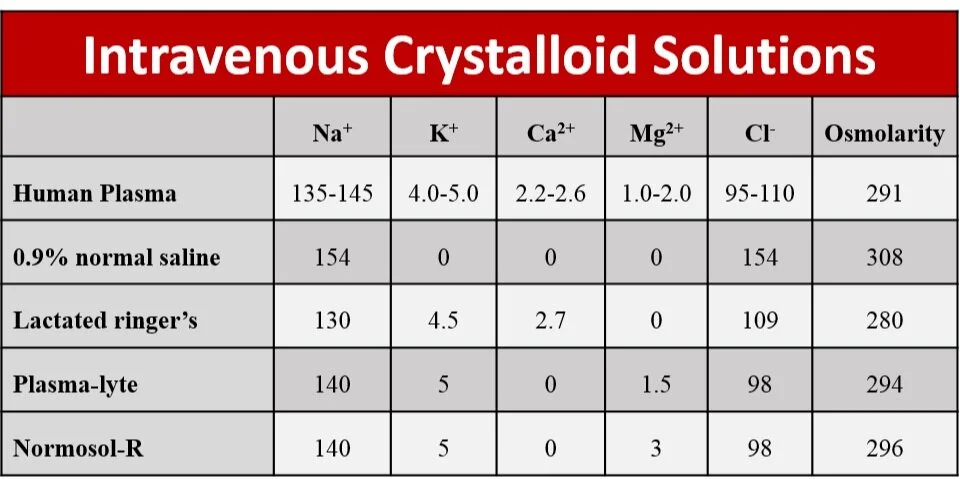
The world of crystalloid is made up of NS and balanced solutions. Balanced solutions, which are designed to more closely reflect the ionic composition and pH of human plasma, are also referred to as chloride restrictive, or buffered, crystalloids. The most common varieties of balanced solutions are lactated Ringer’s (LR), Plasma-Lyte A (PL), and Normosol-R. The composition of these fluids is strikingly different from NS (see Figure 1). Historically, due to a combination of availability, pricing, and precedent, NS has been the default resuscitative fluid in the majority of health care settings. However, observational data has linked NS to increased incidence of acute kidney injury (7), renal replacement therapy, post-operative complications (8), and even mortality (9, 10) when compared to buffered solutions.
Mounting retrospective and observational data prompted large scale, randomized, prospective trials to evaluate the effects of fluid choice on outcomes. Most of these trials were focused in the realm of critical care and demonstrated no significant difference in incidence of AKI (11), or mortality (11, 12, 13). A study published in 2018 by the SALT-ED investigators served as the first prospective randomized trial of fluid resuscitation in the ED (14). Comprised of more than 13,000 subjects, this study demonstrated no difference in hospital-free days between patients admitted to non-ICU settings who were randomized to receive chloride restrictive fluids versus NS. However, patients receiving NS were more likely to die, receive new renal replacement therapy, or suffer from persistent renal dysfunction within 30 days. These outcomes defined the major adverse kidney event (MAKE-30) composite endpoint. These findings were echoed in the concurrently published SMART trial, which demonstrated an increased incidence of MAKE-30 among patients admitted from the ED to the ICU resuscitated with NS compared to buffered crystalloid (13). The authors of each study noted that these findings are hypothesis-generating, given that each was designed primarily to assess mortality rather than the composite outcome. Some critics argue that further large-scale investigation of IVF for all comers is not warranted, and that clinicians should individualize fluid choice based on pathophysiology (15). Fortunately, there are a number of previously published investigations that shed light on fluid choice in a number of common illness states.
Figure 1: intravenous fluid electrolyte concentrations
CONSIDERATIONS IN PATIENT POPULATIONS
SEPSIS:
Fluid administration has been an integral piece of emergent sepsis management since the advent of early goal directed therapy (16). While some components of Dr. Rivers’ protocol have faded, aggressive fluid resuscitation remains a cornerstone of treatment. Subgroup analysis of patients with severe sepsis or septic shock in the Saline versus Albumin Fluid Evaluation (SAFE) study demonstrated a non-significant lower odds of mortality in patients receiving albumin (aOR 0.71, 95% CI 0.52-0.97) (17). However, albumin has not been shown to be superior to crystalloid in subsequent trials, and given the high price associated, it is not recommended for routine use (18). High quality data guiding the choice between crystalloids in sepsis are lacking. A small, retrospective review of 115 septic patients receiving balanced fluids or NS in the ED demonstrated that significantly fewer patients in the balanced group died during their hospitalization (19). In patients receiving both, each increase of 1% in the proportion of balanced fluid was associated with a 2.3% decrease in the odds of in-hospital mortality. Further study is certainly warranted to determine the true effect size of balanced resuscitation in patients with sepsis, a disease process that certainly continues to fill the beds in the ED worldwide.
DIABETIC KETOACIDOSIS:
Patients suffering from diabetic ketoacidosis (DKA) receive aggressive fluid replacement early in their clinical course, due in part to the osmotic diuresis of severe hyperglycemia. Professional society guidelines recommend NS as the initial fluid for restoring intravascular volume (20). LR and PL have been compared to NS in several small studies over the past decade. In a retrospective trial in Australian ICUs, nine patients in the PL-only group had quicker time to resolution of acidemia and less hyperchloremia than the fourteen patients in the NS-only group (21). Similarly, in a prospective, randomized, double blinded trial of NS versus LR in DKA, the LR arm returned to a normal pH more quickly than the NS arm, although this did not reach statistical significance (22). Further, a third prospective, randomized study demonstrated that patients receiving balanced crystalloid were significantly less likely to develop hyperchloremic metabolic acidosis than patients resuscitated with NS (23). The absence of methodologically rigorous studies precludes strong recommendations for balanced resuscitation over NS, but these data serve as a foundation for strong consideration on a case-to-case basis.
HYPERKALEMIA:
Unlike NS, LR and PL both contain potassium. At first glance, supplying a patient afflicted with hyperkalemia more potassium in his or her IVF may seem like a problem. However, consideration of potassium ion exchange and affect of acid-base buffers suggests otherwise. By definition, serum potassium is greater than 5-5.5 millimoles per deciliter in hyperkalemia. Adding any fluid with a concentration less than the serum potassium is unlikely to lead to an increase in the serum value for several reasons. First, adding a hypotonic solution to a fixed volume does not increase tonicity. Second, the pH of the supplied fluid has an effect on serum potassium, with relatively acidic solutions pulling potassium out of the intracellular space, and alkalizing fluid pushing the ion into the cells. Based on these premises, one might expect balanced fluids to decrease serum potassium when compared to NS. Several studies in populations at high risk of hyperkalemia, primarily end-stage renal disease, have demonstrated NS tends to lead to more hyperkalemia (24-26). These trials were all relatively small and in a selected population, raising concerns about generalizability. That said, the argument of biological plausibility is strong enough that hyperkalemia should not represent a strong contraindication to potassium-containing fluids in the majority of patients.
TRAUMATIC BRAIN INJURY:
While patients suffering from critical illness, sepsis, DKA, or hyperkalemia should likely be treated with balanced resuscitation, traumatic brain injury (TBI) represents a different problem all together. The complex interplay of the neurovascular cellular system (consisting of the endothelium and surrounding support cells of the central nervous system) with plasma components is key to maintaining cerebral water volume (27). Hypotonic fluids can have multiplied effects on a disrupted blood brain barrier, thereby increasing the likelihood of developing cerebral edema. In a post-hoc analysis of the SAFE study, patients with TBI were more likely to die when treated with albumin rather than NS (28). While there are no large-scale trials pitting NS against balanced solutions, a retrospective comparison of prehospital fluid for patients with TBI demonstrated an increased adjusted risk for mortality at 30 days for patients receiving LR compared to NS (29). Prospective trials may be forth coming, but for the time being, the physiologic argument against administering hypotonic fluid to patients with significant intracranial injury is enough to justify the use of normal saline in this patient population.
SUMMARY
NS has taken hold as the de facto resuscitative fluid in the majority of health care institutions in the developed world. Recent data suggest that balanced fluids may provide benefit to patients suffering from a wide array of critical illnesses. Outside of TBI, chloride restrictive resuscitation should be the primary strategy in the modern ED.
AUTHORED BY CHRISTOPHER SHAW, MD
Dr. Shaw is a third-year Emergency Medicine resident at the University of Cincinnati
FACULTY EDITORS ANNALS OF B POD EDITORS
Current Editorial Faculty and Articles can be found here
References:
Glassford NJ, Bellomo R. The complexities of intravenous fluid research: questions of scale, volume, and accumulation. Korean J Crit Care Med 2016;31:276–299.
Myburgh, J. A., Finfer, S., Bellomo, R., Billot, L., Cass, A., Gattas, D., ... & McGuinness, S. (2012). Hydroxyethyl starch or saline for fluid resuscitation in intensive care. New England Journal of Medicine, 367(20), 1901-1911.
Perner, A., Haase, N., Guttormsen, A. B., Tenhunen, J., Klemenzson, G., Åneman, A., ... & Bendtsen, A. (2012). Hydroxyethyl starch 130/0.42 versus Ringer's acetate in severe sepsis. New England Journal of Medicine, 367(2), 124-134.
SAFE Study Investigators. (2004). A comparison of albumin and saline for fluid resuscitation in the intensive care unit. New England Journal of Medicine, 350(22), 2247-2256.
Caironi, P., Tognoni, G., Masson, S., Fumagalli, R., Pesenti, A., Romero, M., ... & Iapichino, G. (2014). Albumin replacement in patients with severe sepsis or septic shock. New England Journal of Medicine, 370(15), 1412-1421.
Annane, D., Siami, S., Jaber, S., Martin, C., Elatrous, S., Declère, A. D., ... & Trouillet, J. L. (2013). Effects of fluid resuscitation with colloids vs crystalloids on mortality in critically ill patients presenting with hypovolemic shock: the CRISTAL randomized trial. Jama, 310(17), 1809-1817.
Weinberg, L., Li, M. H. G., Churilov, L., Armellini, A., Gibney, M., Hewitt, T., ... & Bellomo, R. (2018). Associations of fluid amount, type, and balance and acute kidney injury in patients undergoing major surgery. Anaesthesia and intensive care, 46(1), 79-87.
Shaw, A. D., Bagshaw, S. M., Goldstein, S. L., Scherer, L. A., Duan, M., Schermer, C. R., & Kellum, J. A. (2012). Major complications, mortality, and resource utilization after open abdominal surgery: 0.9% saline compared to Plasma-Lyte. Annals of surgery, 255(5), 821-829.
Shaw, A. D., Raghunathan, K., Peyerl, F. W., Munson, S. H., Paluszkiewicz, S. M., & Schermer, C. R. (2014). Association between intravenous chloride load during resuscitation and in-hospital mortality among patients with SIRS. Intensive care medicine, 40(12), 1897-1905.
Raghunathan, K., Bonavia, A., Nathanson, B. H., Beadles, C. A., Shaw, A. D., Brookhart, M. A., ... & Lindenauer, P. K. (2015). Association between initial fluid choice and subsequent in-hospital mortality during the resuscitation of adults with septic shock. Anesthesiology: The Journal of the American Society of Anesthesiologists, 123(6), 1385-1393.
Young, P., Bailey, M., Beasley, R., Henderson, S., Mackle, D., McArthur, C., ... & Reddy, S. (2015). Effect of a buffered crystalloid solution vs saline on acute kidney injury among patients in the intensive care unit: the SPLIT randomized clinical trial. Jama, 314(16), 1701-1710.
Semler, M. W., Wanderer, J. P., Ehrenfeld, J. M., Stollings, J. L., Self, W. H., Siew, E. D., ... & Rice, T. W. (2017). Balanced crystalloids versus saline in the intensive care unit. The SALT randomized trial. American journal of respiratory and critical care medicine, 195(10), 1362-1372.
Semler, M. W., Self, W. H., Wanderer, J. P., Ehrenfeld, J. M., Wang, L., Byrne, D. W., ... & Guillamondegui, O. D. (2018). Balanced crystalloids versus saline in critically ill adults. New England Journal of Medicine, 378(9), 829-839.
Self, W. H., Semler, M. W., Wanderer, J. P., Wang, L., Byrne, D. W., Collins, S. P., ... & Shaw, A. D. (2018). Balanced crystalloids versus saline in noncritically ill adults. New England Journal of Medicine, 378(9), 819-828.
Vincent, J. L., & De Backer, D. (2016). Saline versus balanced solutions: are clinical trials comparing two crystalloid solutions really needed?. Critical Care, 20(1), 250.
Rivers, E., Nguyen, B., Havstad, S., Ressler, J., Muzzin, A., Knoblich, B., ... & Tomlanovich, M. (2001). Early goal-directed therapy in the treatment of severe sepsis and septic shock. New England Journal of Medicine, 345(19), 1368-1377.
SAFE Study Investigators. (2011). Impact of albumin compared to saline on organ function and mortality of patients with severe sepsis. Intensive care medicine, 37(1), 86-96.
Caironi, P., Tognoni, G., Masson, S., Fumagalli, R., Pesenti, A., Romero, M., ... & Iapichino, G. (2014). Albumin replacement in patients with severe sepsis or septic shock. New England Journal of Medicine, 370(15), 1412-1421.
Sethi, M., Owyang, C. G., Meyers, C., Parekh, R., Shah, K. H., & Manini, A. F. (2018). Choice of resuscitative fluids and mortality in emergency department patients with sepsis. The American journal of emergency medicine, 36(4), 625-629.
Kitabchi, A. E., Umpierrez, G. E., Miles, J. M., & Fisher, J. N. (2009). Hyperglycemic crises in adult patients with diabetes. Diabetes care, 32(7), 1335-1343.
Chua, H. R., Venkatesh, B., Stachowski, E., Schneider, A. G., Perkins, K., Ladanyi, S., ... & Bellomo, R. (2012). Plasma-Lyte 148 vs 0.9% saline for fluid resuscitation in diabetic ketoacidosis. Journal of critical care, 27(2), 138-145.
Van Zyl, D. G., Rheeder, P., & Delport, E. (2011). Fluid management in diabetic-acidosis—Ringer's lactate versus normal saline: a randomized controlled trial. QJM: An International Journal of Medicine, 105(4), 337-343.
Mahler, S. A., Conrad, S. A., Wang, H., & Arnold, T. C. (2011). Resuscitation with balanced electrolyte solution prevents hyperchloremic metabolic acidosis in patients with diabetic ketoacidosis. The American journal of emergency medicine, 29(6), 670-674.
Weinberg, L., Harris, L., Bellomo, R., Ierino, F. L., Story, D., Eastwood, G., ... & Mount, P. F. (2017). Effects of intraoperative and early postoperative normal saline or Plasma-Lyte 148® on hyperkalaemia in deceased donor renal transplantation: a double-blind randomized trial. BJA: British Journal of Anaesthesia, 119(4), 606-615.
Khajavi, M. R., Etezadi, F., Moharari, R. S., Imani, F., Meysamie, A. P., Khashayar, P., & Najafi, A. (2008). Effects of normal saline vs. lactated ringer's during renal transplantation. Renal failure, 30(5), 535-539.
O’Malley, C. M., Frumento, R. J., Hardy, M. A., Benvenisty, A. I., Brentjens, T. E., Mercer, J. S., & Bennett-Guerrero, E. (2005). A randomized, double-blind comparison of lactated Ringer’s solution and 0.9% NaCl during renal transplantation. Anesthesia & Analgesia, 100(5), 1518-1524.
Ertmer, C., & Van Aken, H. (2014). Fluid therapy in patients with brain injury: what does physiology tell us?.
SAFE Study Investigators. (2007). Saline or albumin for fluid resuscitation in patients with traumatic brain injury. New England Journal of Medicine, 357(9), 874-884.
Rowell, S. E., Fair, K. A., Barbosa, R. R., Watters, J. M., Bulger, E. M., Holcomb, J. B., ... & PROMMTT Study Group. (2016). The impact of pre-hospital administration of lactated ringer's solution versus normal saline in patients with traumatic brain injury. Journal of neurotrauma, 33(11), 1054-1059.