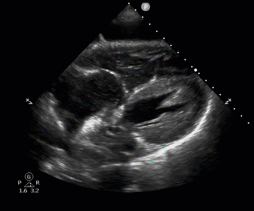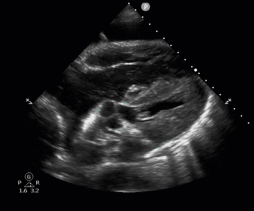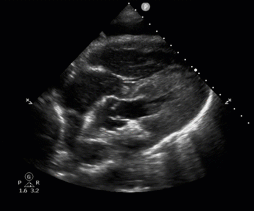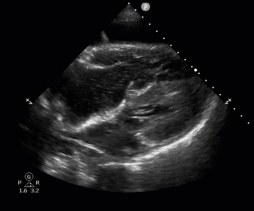
POCUS in Cardiac Arrest
/Out-of-hospital cardiac arrest (OHCA) remains a significant cause of morbidity and mortality worldwide, with over 300,000 estimated annual cases occurring in the United States alone.[1] Despite advances in mechanical chest compression devices, improvement in education and implementation of bystander cardiopulmonary resuscitative (CPR) efforts, and robust pre-hospital care, overall survival rates remain poor at <10%. [2] Current research studies have shifted the focus to identifying prognostic factors to determine which patients are at increased likelihood of achieving return of spontaneous circulation (ROSC) from those that may justify earlier termination of resuscitation. Thus, as point-of-care ultrasound (POCUS) has emerged as a crucial imaging technique to assist in the clinical management of critically ill patients in the Emergency Department (ED), echocardiography-integrated advanced cardiac life support (ACLS) has been brought to the forefront to assist in managing cardiac arrest patients.
POCUS and Diagnosis in Cardiac Arrest
While the focus of cardiac arrest management with ventricular fibrillation and pulseless ventricular tachycardia is to treat the underlying pathologic arrhythmia, patients who present with a non-shockable rhythm rely on the prompt evaluation and management of reversible causes of the arrest. [2] Per the Cardiac Arrest Registry to Enhance Survival, over 80% of OHCA are attributable to a non-shockable rhythm, which can range from asystole, pulseless electrical activity (PEA) with cardiac activity, and PEA without cardiac activity. [2] A study of fifty cardiac arrest patients noted that an intervention was performed in 12% of cases as a direct result of POCUS, demonstrating the impact ultrasound can have in cardiac arrest management by encouraging crucial interventions outside of the ACLS algorithm. [3]
The presence of cardiac activity in PEA arrest is associated with an improved prognosis. Wu et.al. demonstrated that patients in PEA with cardiac activity were 4.35 times more likely to experience ROSC than those without cardiac activity.[4] However, it is very difficult to differentiate between the two PEA states without ultrasound; PEA with cardiac activity has severely insufficient cardiac function, and this state can be equated to a final stage of shock. There may not be a palpable pulse due to the profound nature of the shock, suggesting that PEA with cardiac activity may be inappropriately categorized as PEA without cardiac activity if POCUS is not utilized, losing the subtlety of the diagnosis and difference in prognosis. The complete mechanical dissociation from cardiac electrical impulse in PEA without cardiac activity is a less readily reversible state, and therefore POCUS should be used to quickly differentiate between the two PEA states and assist in patient resuscitation. [5,6]
POCUS and Cardiac Tamponade
Ultrasound can aid in diagnosing reversible causes of cardiac arrest including pericardial effusion and cardiac tamponade, pulmonary embolus, tension pneumothorax, myocardial infarction, and hypovolemia, amongst other etiologies. [3,7-11] The prevalence of pericardial effusion and presumed cardiac tamponade in cardiac arrest has shown to vary widely from 4-40%, however the prognosis for these patients is generally improved from the cardiac arrest population as a whole. One prospective observational study published in 2018 included 177 patients presenting in cardiac arrest, and of the 4% of patients who had cardiac tamponade, ROSC was achieved in 25% of them after a pericardiocentesis. [8] The REASON trial identified pericardial effusion in 34 out of 793 cardiac arrest patients (4%), with an overall survival rate to hospital discharge of 15.4%, significantly higher than the 1.3% overall survival rate for all cardiac arrest patients. [7]
POCUS and Right Heart Strain
Pulmonary embolus is the suspected cause of cardiac arrest in approximately 5% of patients. [12] Many current protocols encourage physicians to evaluate for a dilated right ventricle with a flattened left ventricular chamber as these findings are often sonographically indicative of increased pressures in the pulmonary vasculature, suggesting that a pulmonary embolus may be present. The REASON trial noted that patients with the aforementioned findings had a 6.7% survival rate to hospital discharge after thrombolytics and other appropriate interventions were administered for suspected massive pulmonary embolus. [7]
However, several small studies have raised the concern that not all right heart strain and dilated right ventricles are indicative of a pulmonary embolus,13-19 and that treating all patients with these findings may confer higher risk than true benefit. After inducing cardiac arrest in thirty porcine models, Aagaard et.al. noted that the right ventricle was sonographically dilated from subject baseline during all untreated cardiac arrests secondary to hypovolemia, hyperkalemia, and primary arrhythmia. In fact, the right ventricle was more profoundly enlarged in the hypovolemic group than the hyperkalemic and primary arrhythmia groups. In contrast, the left ventricle was smaller during the arrest compared to the baseline in all groups, thus likely increasing the appearance of right ventricular dilation even more so. [14] Aagaard et.al. completed an additional study including a cohort of 24 pigs comparing induced cardiac arrest from hypoxia, pulmonary embolus, and primary arrhythmia, and again noted right ventricular dilation from baseline in all three groups. Although the models with pulmonary emboli had a more significant dilation of the right ventricle when compared to the other two groups, physicians with basic training in focused cardiac ultrasonography only had modest diagnostic accuracy in detecting such differences. [15] Furthermore, a majority of cardiac arrest patients may continue to have abnormal right ventricular function in the 24 hours after achieving ROSC regardless of the underlying inciting etiology. [13] Researchers hypothesize that the lower-pressure system of the right heart becomes overwhelmed with the lack of forward cardiac output during arrest, and venous pooling leads to acute dilation of the right heart. [13,17] This indicates that right ventricular dilation and other findings of right heart strain may be inherent to cardiac arrest regardless of the underlying etiology, indicating that caution must be used when interpreting these findings and creating a treatment plan during intra-arrest and post-arrest care.
Performing POCUS in Cardiac Arrest
POCUS is traditionally performed during pauses in CPR for pulse and rhythm checks in order to ensure chest compressions are not interrupted. However, recent prospective cohort studies have demonstrated that pauses in CPR are prolonged when POCUS is implemented during a resuscitation. [20,21] Given that maintaining high quality and continuous chest compressions is paramount for maximizing the likelihood of achieving ROSC, there is further work to be done to ensure that POCUS can be incorporated successfully and without unintentional risk to the resuscitation. [22,23] Studies as outlined below have demonstrated that implementation of a standardized approach to POCUS use in cardiac arrest can lead to improvement in time to reach clinical diagnosis and reduction in CPR delays.
What are you looking for?
Several protocols have been proposed in the past decade to standardize and systematically assess for the reversible causes of cardiac arrest. The Cardiac Arrest Sonographic Assessment (CASA) consists of three POCUS exams to occur at serial pulse checks, each evaluating a separate pathology: cardiac tamponade, pulmonary embolus, and cardiac activity. [24] Delays in CPR pauses were reduced by 4.0 seconds when comparing pre- and post-intervention implementation. [25] Other protocols, such as the CAUSE, PEA, US-CAB, FEER/FEEL, and SESAME* have emerged with varied approaches for evaluating cardiac, pulmonary, and abdominal pathologies. [8,10,25-29] While a holistic POCUS examination is ideal in all cardiac arrest patients, it is often not practical in the ED setting due to insufficient time and resources. Therefore, prioritizing cardiac windows is most efficient as the majority of out-of-hospital and in-hospital cardiac arrests are secondary to a primary cardiac etiology. [30-32]
Although outside the scope of this particular post, pulmonary and abdominal views can be obtained when clinical suspicion arises for non-cardiac etiologies of the arrest. A lack of lung sliding can be indicative of a tension pneumothorax; abdominal aortic aneurysm for a rupture; positive focused assessment with sonography for trauma (FAST) exam for hypovolemia secondary to hemorrhage; deep venous thrombosis for presumed pulmonary embolus; along with several other findings.
*CAUSE (Cardiac Arrest Ultra-Sound Exam); PEA (Pulmonary, Epigastric, and Abdominal scans); US-CAB (Ultra-Sound Circulation, Airway, Breathing); FEER/FEEL (Focused Echocardiographic Evaluation in Life support); SESAME (Sequential Echographic Scanning Assessing Mechanism).
When are you looking for it?
POCUS should be implemented as early as feasible to rapidly evaluate for intervenable, reversible causes of cardiac arrest without interrupting the initiation of the resuscitation. A prospective observational study published in 2018 found that the quality of transthoracic echocardiograph (TTE) images obtained during rhythm analysis was, unsurprisingly, superior to the quality of those obtained during chest compressions. [33] Thus, POCUS should be completed during pulse checks to ensure efficiency and success in image acquisition. Placing the probe on the chest in the appropriate position and optimizing depth and gain prior to the pause in chest compressions can also assist in shorter CPR pause durations. [25]
The concept of POCUS reassessment during cardiac arrest has not been robustly studied to date.
A prospective observational study from 2016 enrolled 48 cardiac arrest patients who had a TTE completed upon initial presentation and then serially afterwards, timed every two minutes in coordination with pulse checks. The authors found that, of the 18 patients with sustained cardiac standstill on serial TTE for at least ten minutes, none achieved ROSC. Based on this study, the specificity of predicting eventual non-ROSC with cardiac standstill present for at least ten minutes was 100% with a positive predictive value of 100%.34 Thus, continued reassessment of cardiac activity throughout the resuscitation may be used in conjunction with other predictors of poor prognosis to assist in further management. Repeat POCUS, especially when there is a change in clinical status or after an intervention is performed, is also paramount to assess for response to therapeutic management, and likely has more prognostic value than a single TTE performed on initial presentation.
Who is looking for it?
Although some studies demonstrate that an ultrasound fellowship-trained POCUS performer is likely more adept at acquiring images and reducing CPR pause duration,25 nonexpert sonographers have continually demonstrated to have satisfactory image acquisition during a cardiac arrest as well. [8,10,25,28,35] In one study, emergency physicians acquired images of diagnostic quality in 96% of cases. [10] Another small-sample, prospective study demonstrated 80% agreement in echocardiography interpretation between nonexpert and echocardiography-expert physicians, with no identifiable cases where interpretation could have led to the inappropriate application of therapy. [28] Lastly, a study published in Annals of Emergency Medicine in 2018 looked across specialties, level of training, and self-rated experience level in sonography to assess the interrater reliability in interpreting short clips of TTE images acquired during cardiac arrest. The authors found that, when controlled for all but one of the above variables at a time, there remained a moderate interrater reliability across all cohorts. [36] This indicates that with the appropriate and standardized training, whether through residency or via targeted education for echocardiography, nonexpert sonographers can learn to generally interpret images at the level of more experienced physicians.
Sonographic images are acquired with less delay when performed by separate personnel from the resuscitationist, [25] which is likely a greater consideration than training level itself when performing POCUS during a cardiac arrest. The separation of the roles allows the sonographer to focus purely on image acquisition and interpretation while the resuscitation leader can concentrate on the overall therapeutic approach.
Where are you looking for it?
The optimal cardiac window during cardiac arrest depends not only on ease of image acquisition, but also on patient positioning, body habitus, and likely underlying etiology. The subcostal view, or subxiphoid view, is oftentimes the first default POCUS view in cardiac arrest as it allows for preparation for image acquisition during active CPR. Technically speaking, it is also associated with a higher success rate of interpretable image acquisition, as defined by an adequate view obtained within the CPR pause; a study of 56 patients with one or more cardiac views obtained during cardiac arrest demonstrated a 95% success rate with the subxiphoid view, followed by 85% with the parasternal view, and 50% with the apical view. However, the subcostal view may not be an adequate view to assess for reversible causes in all comers of cardiac arrest, and therefore subsequent attempts with different cardiac windows should be strongly considered as clinically indicated. Communication between the sonographer and resuscitation leader is crucial to ensure that subsequent POCUS attempts are executed without interruption of chest compressions and other interventions.
POCUS and Prognosis in Cardiac Arrest
Recent literature highlights the use of TTE as a potential valuable tool in the prognostication of patients presenting to the ED with OHCA. [3,4,7,8,37-46] The presence of cardiac activity on TTE correlates with improved odds for ROSC, survival to hospital admission (SHA), and survival to hospital discharge (SHD). [4,37-40,46] A meta-analysis of over 1400 patients presenting in non-traumatic, non-shockable cardiac arrest noted that the presence of cardiac activity, compared to the presence of cardiac standstill, had an odds ratio of 16.9 for ROSC, 10.3 for SHA, and 8.03 for SHD, with an overall positive likelihood ratio of 6.87 and negative likelihood ratio of 0.27 for ROSC. [37]
Despite the above, it is imperative to note that not all patients with cardiac standstill are certain to have a poor outcome. While many smaller studies have concluded that patients with cardiac standstill do not survive to hospital discharge, several larger studies have refuted this finding. The REASON trial, one of the largest multi-center, prospective observational studies completed to date, demonstrated that while the presence of cardiac activity was associated with increased likelihood of SHD (3.8%), there were still 0.6% of patients without cardiac activity that survived to hospital discharge. [7]
Limitations
There continue to be limitations in using echocardiography to prognosticate during cardiac arrest. While cardiac activity has been shown to correlate with increased chance of ROSC, there is significant heterogeneity in the definition of cardiac activity and cardiac standstill in the current literature. Definitions range from coordinated ventricular activity with valvular motion [9,47,48]; visible movement of the myocardium [3,6,7,11,41]; any motion detected from the ventricle, atrium, or valve34,42,45; and variable or undefined movements. [4,21,25,28,43,49] Physicians have difficulty interpreting cardiac activity on POCUS because of this ambiguity; a cross-sectional convenience sample survey of 127 physician sonographers, the majority of whom were emergency medicine-trained with varying degrees of training levels, were shown 15 de-identified clips to assess for variability in interpretation of cardiac standstill among all respondents.36 They demonstrated only moderate interrater agreement across all specialties, training levels, and sonographic skill levels, with clips with more subtle findings, such as valvular movement and weak myocardial contraction, resulting in larger percentages of contradictory interpretations.36 Although it demonstrates that physicians of various training levels are able to interpret POCUS images with moderate interrater-reliability as referenced above, this variation in interpretation can prove to have monumental consequences when used as a sole indicator for prognosis during cardiac arrest.
This indicates that a more standardized definition is warranted to guide further research on the use of POCUS in prognostication efforts before it is clinically used to assist in termination of resuscitation in cardiac arrest patients.
In addition, several other limitations in the current literature preclude ED providers from making any definitive conclusions on the use of POCUS to prognosticate in cardiac arrest [4,37-40,46]:
the data is largely based on primary literature with small sample sizes;
the results are difficult to externally validate given the lack of standardized protocols used from study to study;
the training level of the POCUS performer, when defined, is variable;
the timing of POCUS performed during cardiac arrest is heterogenous;
the diagnostic cardiac views obtained vary, often within a study, with minimal explanation
Current research also raises the concern of introducing clinical bias when using echocardiography in cardiac arrest resuscitation. A study from the SHoC-ED investigators published in 2019 demonstrated that patients with cardiac activity on POCUS had longer mean resuscitation times and higher rates of interventions performed, including intubation and epinephrine administration, when compared to those with no cardiac activity on POCUS or when no POCUS was performed.47 And while patients with cardiac activity were noted to have increased rates of ROSC, SHA, and SHD when compared to patients with no cardiac activity, there was no SHA or SHD advantage in those with cardiac activity when compared to those with no POCUS performed. Although difficult to interpret a retrospective review in a conclusive light, this raises the concern that POCUS may bias clinician management of cardiac arrest patients and lead to earlier termination of resuscitation and less life-saving interventions performed despite lack of data to support its use in this way. In fact, there is no data demonstrating that POCUS use in cardiac arrest directly improves patient outcome, such as favorable neurologic outcome post-ROSC. Therefore, further research is required to assess for its effect on clinical patient outcomes as a whole.
Thus, while preliminary research suggests that the use of POCUS to identify cardiac activity in non-traumatic arrest patients may distinguish between patients who are likely to achieve ROSC from those that may be candidates for earlier termination of resuscitation, further research is required to extrapolate this data into clinical practice.
Summary
Echocardiography can be a powerful tool in the rapid evaluation and management of reversible causes of non-traumatic, non-shockable cardiac arrest. Preliminary literature remains promising in the utility of echocardiography as a prognostic tool in cardiac arrest to identify patients with higher likelihood of achieving ROSC with cardiac activity present, or those that may benefit from earlier termination of resuscitation with cardiac standstill. However, at this time, the evidence is not robust enough to support POCUS use as a single prognosticating factor to terminate resuscitative efforts.
Post by Sim Mand, MD (@mand_sim)
Dr. Mand is an Assistant Professor of Emergency Medicine at the University of Wisconsin and prior US Fellow and Chief Resident at the University of Cincinnati
Peer Editing by Lori Stolz, MD RDMS (@sonostolz)
Dr. Stolz is the Division Director of Ultrasound at the University of Cincinnati
References
1. Virani SS, Alonso A, Benjamin EJ, et al. Heart disease and stroke statistics-2020 update: A report from the american heart association. Circulation. 2020;141(9):e139-e596. doi: 10.1161/CIR.0000000000000757 [doi].
2. Cardiac arrest registry to enhance survival. https://mycares.net.
3. Hayhurst C, Lebus C, Atkinson PR, et al. An evaluation of echo in life support (ELS): Is it feasible? what does it add? Emerg Med J. 2011;28(2):119-121. doi: 10.1136/emj.2009.084202 [doi].
4. Wu C, Zheng Z, Jiang L, et al. The predictive value of bedside ultrasound to restore spontaneous circulation in patients with pulseless electrical activity: A systematic review and meta-analysis. PLoS One. 2018;13(1):e0191636. doi: 10.1371/journal.pone.0191636 [doi].
5. Mehta C, Brady W. Pulseless electrical activity in cardiac arrest: Electrocardiographic presentations and management considerations based on the electrocardiogram. American Journal of Emergency Medicine. 2012;30(1):236-239.
6. Zengin s, Yavuz E, Al B, et al. Benefits of cardiac sonography performed by a non-expert monographer in patients with non-traumatic cardiopulmonary arrest. Resuscitation. 2016:105-109.
7. Gaspari R, Weekes A, Adhikari S, et al. Emergency department point-of-care ultrasound in out-of-hospital and in-ED cardiac arrest. Resuscitation. 2016;109:33-39.
8. Lien WC, Hsu SH, Chong KM, et al. US-CAB protocol for ultrasonographic evaluation during cardiopulmonary resuscitation: Validation and potential impact. Resuscitation. 2018;127:125-131. doi: S0300-9572(18)30061-3 [pii].
9. Chardoli M, Heidari F, Rabiee H, Sharif-Alhoseini M, Shokoohi H, Rahimi-Movaghar V. Echocardiography integrated ACLS protocol versus conventional cardiopulmonary resuscitation in patients with pulseless electrical activity cardiac arrest. Chin J Traumatol. 2012;15(5):284-287. doi: S1008-1275(15)30331-X [pii].
10. Breitkreutz R, Walcher F, Seeger FH. Focused echocardiographic evaluation in resuscitation management: Concept of an advanced life support-conformed algorithm. Crit Care Med. 2007;35(5 Suppl):150. doi: 10.1097/01.CCM.0000260626.23848.FC [doi].
11. Tayal VS, Kline JA. Emergency echocardiography to detect pericardial effusion in patients in PEA and near-PEA states
. Resuscitation. 2003;59(3):315-8.
12. Kürkciyan I, Meron G, Sterz F, et al. Pulmonary embolism as a cause of cardiac arrest: Presentation and outcome. Arch Intern Med. 2000;160(10):1529-1535. doi: 10.1001/archinte.160.10.1529 [doi].
13. Wardi G, Blanchard D, Dittrich T, Kaushal K, Sell R. Right ventricle dysfunction and echocardiographic parameters in the first 24h following resuscitation in the post-cardiac arrest patient: A retrospective cohort study. Resuscitation. 2016;103:71-74. doi: S0300-9572(16)00125-8 [pii].
14. Aagaard R, Granfeldt A, Bøtker MT, Mygind-Klausen T, Kirkegaard H, Løfgren B. The right ventricle is dilated during resuscitation from cardiac arrest caused by hypovolemia: A porcine ultrasound study. Crit Care Med. 2017;45(9):e963-e970. doi: 10.1097/CCM.0000000000002464 [doi].
15. Aagaard R, Caap P, Hansson NC, Bøtker MT, Granfeldt A, Løfgren B. Detection of pulmonary embolism during cardiac arrest-ultrasonographic findings should be interpreted with caution. Crit Care Med. 2017;45(7):e695-e702. doi: 10.1097/CCM.0000000000002334 [doi].
16. Steen S, Liao Q, Pierre L, Paskevicius A, Sjöberg T. The critical importance of minimal delay between chest compressions and subsequent defibrillation: A haemodynamic explanation. Resuscitation. 2003;58(3):249-258. doi: S030095720300265X [pii].
17. Berg RA, Sorrell VL, Kern KB, et al. Magnetic resonance imaging during untreated ventricular fibrillation reveals prompt right ventricular overdistention without left ventricular volume loss. Circulation. 2005;111(9):1136-1140. doi: 01.CIR.0000157147.26869.31 [pii].
18. Sørensen AH, Wemmelund KB, Møller-Helgestad OK, Sloth E, Juhl-Olsen P. Asphyxia causes ultrasonographic D-shaping of the left ventricle--an experimental porcine study. Acta Anaesthesiol Scand. 2016;60(2):203-212. doi: 10.1111/aas.12606 [doi].
19. Comess KA, DeRook FA, Russell ML, Tognazzi-Evans TA, Beach KW. The incidence of pulmonary embolism in unexplained sudden cardiac arrest with pulseless electrical activity. Am J Med. 2000;109(5):351-356. doi: S0002-9343(00)00511-8 [pii].
20. Huis In 't Veld, M. A., Allison MG, Bostick DS, et al. Ultrasound use during cardiopulmonary resuscitation is associated with delays in chest compressions. Resuscitation. 2017;119:95-98.
21. Clattenburg EJ, Wroe P, Brown S, et al. Point-of-care ultrasound use in patients with cardiac arrest is associated prolonged cardiopulmonary resuscitation pauses: A prospective cohort study. Resuscitation. 2018;122:65-68.
22. Cheskes S, Schmicker RH, Verbeek PR, et al. The impact of peri-shock pause on survival from out-of-hospital shockable cardiac arrest during the resuscitation outcomes consortium PRIMED trial. Resuscitation. 2014;85(3):336-342. doi: S0300-9572(13)00814-9 [pii].
23. Kellum MJ, Kennedy KW, Barney R, et al. Cardiocerebral resuscitation improves neurologically intact survival of patients with out-of-hospital cardiac arrest. Ann Emerg Med. 2008;52(3):244-252. doi: 10.1016/j.annemergmed.2008.02.006 [doi].
24. Gardner KF, Clattenburg EJ, Wroe P, Singh A, Mantuani D, Nagdev A. The cardiac arrest sonographic assessment (CASA) exam - A standardized approach to the use of ultrasound in PEA. Am J Emerg Med. 2018;36(4):729-731.
25. Clattenburg EJ, Wroe PC, Gardner K, et al. Implementation of the cardiac arrest sonographic assessment (CASA) protocol for patients with cardiac arrest is associated with shorter CPR pulse checks. Resuscitation. 2018;131:69-73.
26. Testa A, Cibinel GA, Portale G, et al. The proposal of an integrated ultrasonographic approach into the ALS algorithm for cardiac arrest: The PEA protocol. Eur Rev Med Pharmacol Sci. 2010;14(2):77-88.
27. Hernandez C, Shuler K, Hannan H, Sonyika C, Likourezos A, Marshall J. C.A.U.S.E.: Cardiac arrest ultra-sound exam--a better approach to managing patients in primary non-arrhythmogenic cardiac arrest. Resuscitation. 2008;76(2):198-206. doi: S0300-9572(07)00420-0 [pii].
28. Niendorff DF, Rassias AJ, Palac R, Beach ML, Costa S, Greenberg M. Rapid cardiac ultrasound of inpatients suffering PEA arrest performed by nonexpert sonographers. Resuscitation. 2005;67(1):81-87. doi: S0300-9572(05)00194-2 [pii].
29. Lichtenstein DA. How can the use of lung ultrasound in cardiac arrest make ultrasound a holistic discipline. the example of the SESAME-protocol. Med Ultrason. 2014;16(3):252-255. doi: 10.11152/mu.2013.2066.163.dal1 [doi].
30. Chen N, Callaway CW, Guyette FX, et al. Arrest etiology among patients resuscitated from cardiac arrest. Resuscitation. 2018;130:33-40. doi: S0300-9572(18)30295-8 [pii].
31. Bergum D, Haugen BO, Nordseth T, Mjølstad OC, Skogvoll E. Recognizing the causes of in-hospital cardiac arrest--A survival benefit. Resuscitation. 2015;97:91-96. doi: S0300-9572(15)00810-2 [pii].
32. Wallmuller C, Meron G, Kurkciyan I, Schober A, Stratil P, Sterz F. Causes of in-hospital cardiac arrest and influence on outcome. Resuscitation. 2012;83(10):1206-1211. doi: 10.1016/j.resuscitation.2012.05.001 [doi].
33. Aagaard R, Lofgren B, Grofte T, et al. Timing of focused cardiac ultrasound during advanced life support - A prospective clinical study. Resuscitation. 2018;124:126-131. doi: S0300-9572(17)30789-X [pii].
34. Kim HB, Such JY, Choi JH, Cho YS. Can serial focussed echocardiographic evaluation in life support (FEEL) predict resuscitation outcome or termination of resuscitation (TOR)? A pilot study. Resuscitation. 2016:21-6.
35. Flato UA, Paiva EF, Carballo MT, Buehler AM, Marco R, Timerman A. Echocardiography for prognostication during the resuscitation of intensive care unit patients with non-shockable rhythm cardiac arrest. Resuscitation. 2015;92:1-6. doi: 10.1016/j.resuscitation.2015.03.024 [doi].
36. Hu K, Gupta N, Teran F, Saul T, Nelson BP, Andrus P. Variability in interpretation of cardiac standstill among physician sonographers. Annals of Emergency Medicine. 2018;71(2):193-198. http://dx.doi.org/10.1016/j.annemergmed.2017.07.476. doi: 10.1016/j.annemergmed.2017.07.476.
37. Lalande E, Burwash-Brennan T, Burns K, et al. Is point-of-care ultrasound a reliable predictor of outcome during atraumatic, non-shockable cardiac arrest? A systematic review and meta-analysis from the SHoC investigators. Resuscitation. 2019;139:159-166.
38. Long B, Alerhand S, Maliel K, Koyfman A. Echocardiography in cardiac arrest: An emergency medicine review. Am J Emerg Med. 2018;36(3):488-493.
39. Kedan I, Ciozda W, Palatinus JA, Palatinus HN, Kimchi A. Prognostic value of point-of-care ultrasound during cardiac arrest: A systematic review. Cardiovasc Ultrasound. 2020;18(1):1-8. doi: 10.1186/s12947-020-0185-8 [doi].
40. Tsou PY, Kurbedin J, Chen YS, et al. Accuracy of point-of-care focused echocardiography in predicting outcome of resuscitation in cardiac arrest patients: A systematic review and meta-analysis. Resuscitation. 2017;114:92-99.
41. Ozen C, Salcin E, Akoglu H, Onur O, Denizbasi A. Assessment of ventricular wall motion with focused echocardiography during cardiac arrest to predict survival. Turkish Journal of Emergency Medicine. 2016;16(1):12-6.
42. Tomruk O, Erdur B, Cetin G, Ergin A, Avcil M, Kapci M. Assessment of cardiac ultrasonography in predicting outcome in adult cardiac arrest. Journal of International Medical Research. 2012;40(2):804-809. https://journals.sagepub.com/doi/full/10.1177/147323001204000247. doi: 10.1177/147323001204000247.
43. Cebicci H, Salt O, Gurbuz S, Koyuncu S, Bol O. Benefit of cardiac sonography for estimating the early term survival of the cardiopulmonary arrest patients. Hippokratia. 2014;18(2):125-129.
44. Blaivas M, Fox JC. Outcome in cardiac arrest patients found to have cardiac standstill on the bedside emergency department echocardiogram. Acad Emerg Med. 2001;8(6):616-621.
45. Salen P, Melniker L, Chooljian C, et al. Does the presence or absence of sonographically identified cardiac activity predict resuscitation outcomes of cardiac arrest patients? Am J Emerg Med. 2005;23(4):459-462. doi: S0735675705001269 [pii].
46. Blyth L, Atkinson P, Gadd K, Lang E. Bedside focused echocardiography as predictor of survival in cardiac arrest patients: A systematic review. Acad Emerg Med. 2012;19(10):1119-1126.
47. Atkinson PR, Beckett N, French J, Banerjee A, Fraser J, Lewis D. Does point-of-care ultrasound use impact resuscitation length, rates of intervention, and clinical outcomes during cardiac arrest? A study from the sonography in hypotension and cardiac arrest in the emergency department (SHoC-ED) investigators. Cureus. 2019;11(4):e4456.
48. Flato UA, Paiva EF, Carballo MT, Buehler AM, Marco R, Timerman A. Echocardiography for prognostication during the resuscitation of intensive care unit patients with non-shockable rhythm cardiac arrest. Resuscitation. 2015;92:1-6.
49. Huis In'tVeld MA, Allison MG, Bostic DS, et al. Ultrasound use during cardiopulmonary resuscitation is associated with delays in chest compressions. Resuscitation. 2017:95-98.